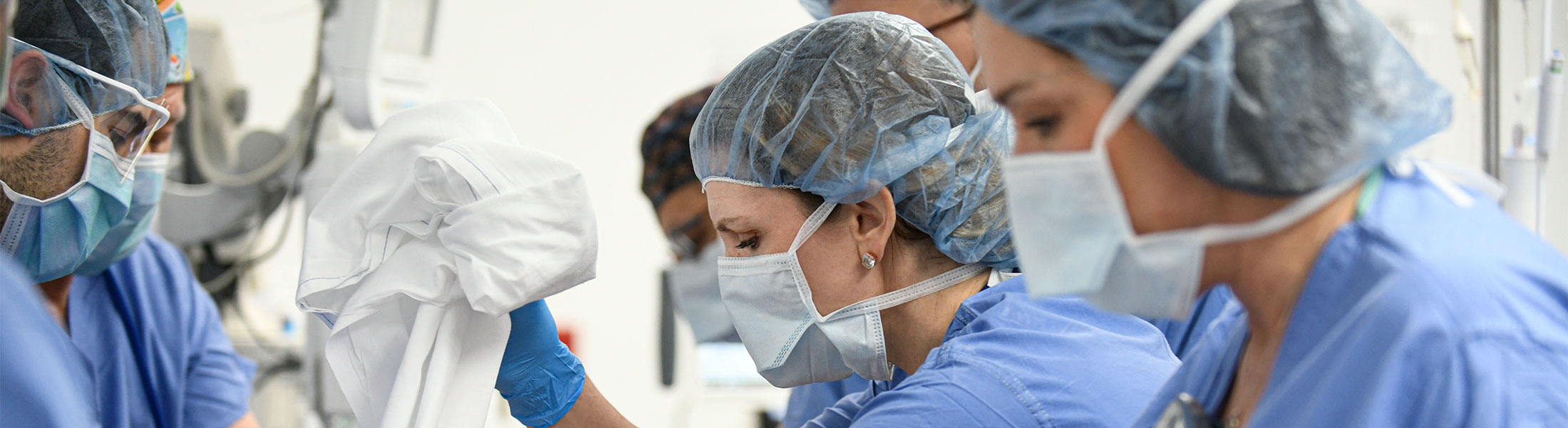
With thousands of health care professionals, hundreds of millions of dollars of cutting-edge technology and some of the brightest medical minds in the world, Kaleida Health is uniquely qualified to provide world class patient care.
Specialty Centers
Our specialty services feature highly trained physicians utilizing the latest technology to treat a variety of conditions and diseases. From cardiac care to maternity to cancer services, patients are assured they are in the best hands possible at a Kaleida Health facility.
Our Hospitals
Buffalo General Medical Center/Gates Vascular Institute
100 High Street
Buffalo, NY 14203
Bradford Regional Medical Center/Olean General Hospital
116 Interstate Parkway
Bradford, PA 16701
Education & Prevention
We believe that the first step toward prevention is education. That is why we take a proactive approach to prevention. We offer a variety of programs and services to the public before they become patients. While physicians, students and staff instill confidence in people to make healthy decisions, everyone benefits from the learning experience with a positive attitude about health care.
Adult
Home Care Services
The talented, experienced nurses of the Visiting Nursing Association (VNA) and the VNA Home Care Services can provide at-home services on a day-to-day basis. The VNA is united in its mission to provide compassionate, high-quality and cost-effective home health care to the Western New York community.
Adult
Laboratory Services
The clinical laboratories of Kaleida Health play a critical role in the care of our patients. Without their expertise, physicians wouldn't know whether a disease or condition exists and to what extent. A patient's treatment plan is in large part based on the findings of the our highly specialized team of laboratory scientists and technicians.
Adult
Long-term Care
Aging is an inevitable process, a continuous period that can be spiritual, joyous and fulfilling. We can provide a comfortable setting complete with required amenities and services for those who need extra care. Our long-term care facilities accommodate short or long-term care with several locations to serve the Western New York area.
Pediatric
Maternity/Women's Services
Women have a special set of needs that change throughout the life cycle. Women's Services at WCHOB provide comprehensive care for any medical concern. From birthing classes to mammograms, we advance the health of women with dedicated service
Adult
Outpatient Centers
Outpatient Centers provide medical care, rehabilitation or tests without the need for an overnight stay in a hospital. In some cases, the experience is similar to a standard visit to your primary care physician. In other cases, testing, rehabilitation or other activities needed for your unique healthcare needs may make your visit a bit longer. Your healthcare provider can give you an estimate of the time you will need to allot for your visit.
Adult
Pediatric
- Allergy/Immunology
- Autism Spectrum Disorder Center
- Cardiology
- Children's Psychiatry Center
- Craniofacial Center of Western New York
- Dermatology
- Down Syndrome Program
- Ear, Nose & Throat
- EEG/Neurophysiology Labs
- Endocrinology/Diabetes
- Gastroenterology & Nutrition
- Genetics (Pediatric and Adult)
- Healthy Weigh
- Hertel Elmwood Medicine Center
- Infectious Diseases
Rehabilitation Services
Kaleida Health Rehabilitation programs are designed to provide comprehensive rehabilitation for adults. Our goal is to help patients maximize their recovery from injury or illness with a combination of individualized physical, occupational and speech therapies.
Adult
Pediatric
Support Services
Many of our most important services happen behind the scenes. Volunteers, human resources, plant operations and safety, just to name a few, are services that make Kaleida Health the premier health care system in Western New York. Their reinforcement, protection and assistance uphold the mission and goals of our organization.
Adult
- Access Center
- Adult Day Services
- Bereavement Services
- Blood Draw
- Breast Reconstruction Surgery Information
- Cancer Rehab & Recovery
- Case Management/Social Work & Discharge Planning
- Childbirth Education
- Community Health Department
- Emergency Medical Services (EMS)
- Family Planning Center
- Financial Assistance Program
- Greater Buffalo United Accountable Healthcare
- Interpreter / Translation and Deaf Services
- Medical Records
- Pastoral Care
- Patient Financial Services
- Security & Safety
- Volunteer Services
Pediatric
- Access Center
- Admissions
- Blood Donation
- Child Life Department
- Childbirth Education
- Financial Assistance Program
- Health Information Management Department (Medical Records)
- Interpreter / Translation and Deaf Services
- Medical Records
- Patient Access Services
- Patient and Family Centered Care
- Patient Financial Services
- Patient Management Services (Discharge Planning and Social Work)
- Pharmacy - Outpatient
- Spiritual Care
- Volunteer Services